11 Protein Misfolding Diseases
Warning: Undefined variable $post in /home/dietofli/public_html/wp-content/plugins/code-snippets/php/snippet-ops.php(584) : eval()'d code on line 3
Warning: Attempt to read property "ID" on null in /home/dietofli/public_html/wp-content/plugins/code-snippets/php/snippet-ops.php(584) : eval()'d code on line 3
The estimated reading time is 4 minutes
Warning: Undefined variable $post in /home/dietofli/public_html/wp-content/plugins/oxygen/component-framework/components/classes/code-block.class.php(115) : eval()'d code on line 3
Warning: Attempt to read property "ID" on null in /home/dietofli/public_html/wp-content/plugins/oxygen/component-framework/components/classes/code-block.class.php(115) : eval()'d code on line 3
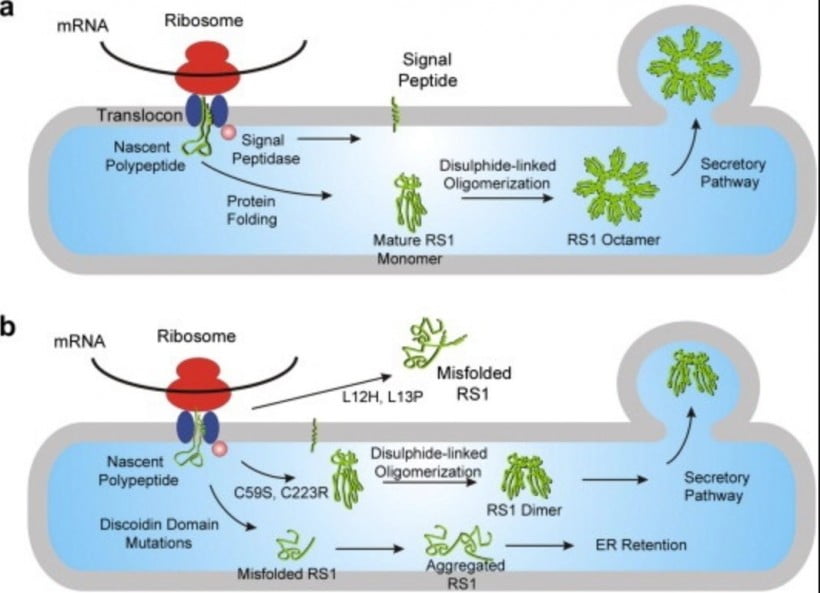
Proteins are complex molecules that play many critical roles within the human body. Many of these roles occur at the cellular level and are undetectable to us as we go about our daily lives—until something goes wrong.
Protein Misfolding Diseases
When proteins unfold improperly or misfold, they can no longer carry out their cellular responsibilities and begin to degenerate. Over time, clumps of degenerated proteins start to aggregate in the cell, causing various types of dysfunction and eventually cell death. Here is a list of diseases that are commonly associated with protein aggregation and its effects on the cells of the body:
- Alzheimer’s disease (AD)—when amyloid and tau proteins unfold in and around brain cells, proteins form plaques and tangles that cause these neurons to lose function. As the neurons degenerate, they become unable to communicate with surrounding cells, affecting memory, language, vision, and more.
- Cystic fibrosis (CF)—mutations within the cystic fibrosis transmembrane regulator gene (CFTR) cause CFTR protein misfolding, rendering these proteins unable to move salts in and out of the cell. As a result, the mucus accumulates within the respiratory, digestive, and reproductive systems, causing damage and, eventually, respiratory failure.
- Creutzfeldt–Jakob disease (CJD)—misfolding of a prion protein can cause protein aggregation and neuron death in humans, leading to a sponge-like lesion in the brain. Humans can either inherit this prion or receive it through transmission, but either form causes severe mental and physical impairment as well as eventual death.
- Familial amyloid polyneuropathy (FAP)—this rare, heritable, progressive disease is caused by mutations in the TTR gene, which provides instructions for a liver protein responsible for transporting vitamin A and hormones. Improper folding causes protein aggregations in the nerves, heart, kidneys, and eyes, causing tingling, high blood pressure, abnormal digestion, and eventually heart disease and death.
- Gaucher’s disease (GD)—this heritable gene mutation causes protein misfolding and degradation of an enzyme that enables the breakdown of fatty substances in the spleen, liver, and bone tissue. GD can cause enlargement of the organs, poor function, bone weakness, and poor clotting.
- Huntington’s disease (HD)—is caused by a defective gene on chromosome 4, HD results when the gene codes for defective huntingtin, which misfolds and aggregates in the cells. Eventually, irreversible degeneration and cell death linked to defective huntingtin leads to mood changes, obsessive-compulsive disorder (OCD), decreased thinking and reasoning, and abnormal involuntary body movements.
- Machado‐Joseph disease (MJD)—also known as spinocerebellar ataxia (SCA), MJD causes progressive clumsy movements of the arms and legs, limb spasticity, slurred speech, and difficulties with vision. MJD results from a mutation in the ATXN gene that codes for ataxin-3 and causes misfolding and aggregation in the cells of the hindbrain.
- Nephrogenic diabetes insipidus (NDI)—mutations in the gene encoding for the precursors of the antidiuretic hormone arginine vasopressin (AVP) cause incompetent folding of the responsible protein. These misfolded proteins can form fibrillar aggregates within the cell’s endoplasmic reticulum (ER), causing cell degradation, increased urine production, and dehydration.
- Parkinson’s disease (PD) is caused by a progressive loss of nerve cells in the part of the brain responsible for dopamine production, enabling cell messaging that allows body movement—PD ultimately leads to tremors, slow movements, and muscle rigidity. Protein aggregates known as Lewy bodies within the cells of the substantia nigra are thought to cause this early cell degradation.
- Retinitis pigmentosa (RP)—mutations in any of the genes that create photoreceptors in the retina can lead to abnormally functioning or toxic proteins. These proteins damage the photoreceptors and lead to cell degradation and death, causing progressive vision loss.
- Senile systemic amyloidosis (SSA)—SSA is caused by abnormal folding and accumulation of the transthyretin (TTR) protein in the organs and heart. The condition mostly affects older men and can cause organ failure, heart rhythm issues, and heart failure.
Identifying the precise pathways of protein dysfunction and disease can help researchers determine potential therapies to slow or halt the progression of the above diseases. In the future, techniques to prevent protein misfolding and aggregation could provide much-needed relief for those diagnosed with protein misfolding diseases.
Resources:
https://medlineplus.gov/genetics/understanding/howgeneswork/protein/
http://amyloidosis.org/facts/wild-type/
https://academic.oup.com/jmcb/article/2/4/180/865830
https://www.parkinson.org/Understanding-Parkinsons/Causes
https://rarediseases.org/rare-diseases/nephrogenic-diabetes-insipidus/
https://www.ninds.nih.gov/disorders/patient-caregiver-education/fact-sheets/machado-joseph-disease-fact-sheet
https://pubmed.ncbi.nlm.nih.gov/22200539/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3819655/
https://fapnewstoday.com/what-is-familial-amyloid-polyneuropathy/
https://www.cdc.gov/prions/cjd/about.html
https://www.mayoclinic.org/diseases-conditions/cystic-fibrosis/symptoms-causes/syc-20353700
https://www.nhs.uk/conditions/alzheimers-disease/causes/














